What is IGF-1 LR3?
IGF-1 LR3 is a synthetically engineered analog of insulin-like growth factor-1 (IGF-1), classified as a recombinant peptide hormone within the insulin/IGF signaling family. It was developed for research purposes through specific structural modifications. Specifically, an arginine substitution at position 3 and a 13-amino-acid N-terminal extension, designed to reduce binding to IGF-binding proteins and prolong interaction with the insulin-like growth factor-1 receptor (IGF-1R).
In research investigations, IGF-1 LR3 is studied as a tool for examining IGF-1R–mediated signaling, cellular growth pathways, and downstream metabolic signaling processes, primarily in vitro and in animal models. Its altered binding characteristics differentiate it from native IGF-1 in experimental settings, allowing researchers to explore receptor dynamics under controlled conditions.
However, controlled human clinical evidence remains limited, so no biological outcomes are implied in human trials. All interpretations should remain within a preclinical research framework. Bluum Peptides supplies IGF-1 LR3 as a high-purity, lyophilized research compound for research purposes only. The material’s purity is tested and verified by Janoshik and other leading labs to help support reproducible laboratory studies. IGF-1 LR3 is not approved for human or veterinary application.
IGF-1 LR3 Mechanism of Action (Research Only)
IGF-1 LR3 is a synthetic peptide analog of insulin-like growth factor-1 that exerts its biological activity primarily through activation of the insulin-like growth factor-1 receptor (IGF-1R), a transmembrane receptor tyrosine kinase.
Current understanding of the IGF-1 LR3 mechanism is derived predominantly from in vitro experiments and non-clinical animal models, where it is used as a tool to study receptor signaling dynamics, cellular growth regulation, and metabolic signaling pathways.
Structural and Chemical Basis
IGF-1 LR3 is a recombinant peptide consisting of approximately 83 amino acids, making it longer than native IGF-1 due to a 13-amino-acid N-terminal extension and an arginine substitution at position three[1]. It is classified as a synthetic peptide hormone analog within the insulin/IGF family.
These structural modifications significantly reduce its affinity for IGF-binding proteins (IGFBPs), a feature that experimentally prolongs receptor availability in research models[2]. As a result, IGF-1 LR3 demonstrates increased stability and sustained receptor interaction in vitro, which is useful for controlled signaling studies rather than reflecting physiological behavior.
IGF-1 Receptor Engagement
In experimental systems, IGF-1 LR3 binds selectively to IGF-1R on the cell surface, initiating receptor autophosphorylation and activation[3]. This interaction is similar in nature to native IGF-1 but differs in duration and binding dynamics due to reduced sequestration by IGFBPs.
Researchers use this property to examine how prolonged receptor activation influences intracellular signaling strength and timing. These studies help clarify how receptor occupancy and signal persistence affect downstream cellular responses in non-clinical settings.
Downstream Signaling Pathways
Following IGF-1R activation, IGF-1 LR3 has been observed to stimulate canonical intracellular signaling cascades such as the PI3K/Akt and MAPK/ERK pathways in research models[4]. These pathways are widely studied for their roles in cellular growth, metabolism, and survival signaling.
In research contexts, IGF-1 LR3 allows investigators to probe how variations in ligand structure influence pathway bias, signal amplification, or cross-talk between signaling networks, without attributing these observations to functional outcomes in humans.
Cellular Growth and Metabolic Signaling Context
Within experimental models, IGF-1 LR3 is frequently used to study mechanisms related to cellular proliferation, protein synthesis signaling, and metabolic regulation at the cellular level[5].
These processes are better understood in terms of energy utilization, cell maintenance, and growth signaling, though they remain mechanistic observations rather than biological effects. Most findings originate from cultured cell lines or animal studies designed to isolate signaling variables under controlled conditions.
IGF-1 LR3 is supplied as a high-purity, research-grade lyophilized compound intended exclusively for research purposes. It serves as a valuable experimental tool for exploring IGF-1R-mediated signaling mechanisms, but no clinical, therapeutic, diagnostic, or human applications are hereby implied.
IGF-1 LR3 Research Applications (Observations from Studies)
IGF-1 LR3 has been investigated in preclinical and limited early-phase research settings as a modified insulin-like growth factor used to study IGF-1 receptor–mediated biology. All information presented here reflects observations from controlled experimental systems, including in vitro studies and animal models, and does not represent established clinical outcomes.
These findings should not be interpreted as extending to human or veterinary use and remain confined to laboratory and research contexts.
Cellular Growth and Proliferation Signaling
In experimental models, IGF-1 LR3 is commonly studied for its role in regulating cellular growth and proliferation through sustained IGF-1 receptor activation[6]. Research has shown directional increases in markers associated with cell cycle progression and protein synthesis signaling in cultured cell lines.
Because IGF-1 LR3 exhibits reduced binding to IGF-binding proteins, researchers use it to examine how prolonged receptor engagement alters growth-related signaling compared to native IGF-1. In simpler terms, it allows scientists to study how cells respond when growth signals persist longer than usual under controlled conditions.
Muscle Cell and Tissue Research Models
IGF-1 LR3 has been utilized in muscle cell cultures and animal models to explore pathways involved in myoblast differentiation and tissue maintenance signaling[7]. Observations include favorable shifts in signaling associated with muscle cell growth and repair mechanisms, although this doesn’t imply any functional or performance outcomes.
Compared to unmodified IGF-1, IGF-1 LR3 is of interest because its extended receptor interaction helps researchers isolate signaling effects without frequent ligand degradation. This makes it a useful comparative tool for studying tissue-specific IGF-1R dynamics in laboratory environments.
Metabolic and Insulin-Related Signaling Studies
In metabolic research, IGF-1 LR3 has been examined for its interactions with insulin-related signaling pathways, particularly where IGF-1R and insulin receptor signaling overlap. Studies in cell and animal models suggest directional modulation of glucose and lipid metabolism signaling cascades, primarily through PI3K/Akt pathway engagement[8].
These observations are used to better understand how IGF-family peptides influence metabolic signaling networks at a molecular level. In accessible terms, researchers study how growth-related signals intersect with energy regulation inside cells.
Comparative Signaling and Pathway Analysis
Researchers also use IGF-1 LR3 as a comparative compound to study differences between transient versus sustained IGF-1 receptor activation. Its structural modifications make it useful for investigating signal duration, pathway bias, and downstream cross-talk compared to native IGF-1 or other growth factor analogs.
This comparative approach is valuable for mechanistic research rather than for establishing superiority or applied outcomes. Such studies help clarify how small molecular changes influence complex signaling behavior in experimental systems.
IGF-1 LR3 vs IGF-1 (Mecasermin) vs Insulin
|
Attribute |
IGF-1 LR3 |
IGF-1 (Mecasermin) |
Insulin |
|
Molecular classification |
Modified IGF-1 analog with N-terminal extension and amino acid substitution |
Recombinant human IGF-1 |
Peptide hormone |
|
Primary receptor targets |
IGF-1 receptor (IGF-1R); reduced affinity for IGF-binding proteins |
IGF-1 receptor (IGF-1R) |
Insulin receptor (IR); limited cross-talk with IGF-1R at high concentrations |
|
Binding characteristics |
Prolonged receptor interaction and extended half-life due to reduced IGFBP binding |
Native IGF-1 binding profile with significant IGFBP interaction |
Rapid receptor binding with short circulating half-life |
|
Mechanism complexity |
Single-pathway growth factor signaling with altered pharmacokinetics |
Single-pathway growth factor signaling |
Single-pathway metabolic signaling |
|
Biological pathway focus |
Cell growth, proliferation, and anabolic signaling models |
Growth and developmental signaling studies |
Glucose uptake, metabolic regulation, and energy balance |
|
Metabolic vs cellular scope |
Primarily cellular and tissue-level growth modeling |
Cellular and systemic growth regulation |
Systemic metabolic regulation |
|
Research stage / regulatory status |
Research-use-only compound |
FDA-approved prescription medication |
FDA-approved prescription medication |
|
Common research applications |
Extended IGF-1 signaling, receptor dynamics, and growth pathway modeling |
Physiological IGF-1 activity and deficiency modeling |
Metabolic control, insulin sensitivity, and signaling studies |
|
Intended use classification |
Laboratory research chemical. Not approved for human or veterinary use. |
Prescription drug. Approved medication; clinical use requires prescription |
Prescription drug. Approved medication; clinical use requires prescription |
Important Notice: Comparisons are provided strictly for research and modeling context, not for clinical equivalence or substitution.
IGF-1 LR3 Laboratory Safety & Handling (Research Use Only)
IGF-1 LR3 is supplied strictly for controlled laboratory research applications. As a modified peptide growth factor, it does not have a fully established toxicological or clinical safety profile. All handling, storage, and disposal considerations apply solely within regulated research environments and should follow institutional, local, and regulatory laboratory standards.
General safety and handling considerations may vary depending on formulation, preparation methods, and experimental protocols. When working with IGF-1 LR3, laboratories typically observe the following high-level best practices:
- Sterile technique: Handle under aseptic conditions where applicable, particularly during reconstitution or transfer, to minimize contamination and preserve experimental integrity.
- Personal protective equipment (PPE): Use appropriate PPE such as laboratory gloves, protective eyewear, and lab coats in accordance with institutional SOPs.
- Engineering controls: Perform weighing, reconstitution, or transfers in suitable containment (e.g., biosafety cabinet or fume hood) to reduce aerosolization or accidental exposure.
- Storage conditions: Store lyophilized material under controlled, low-temperature conditions as specified in product documentation, with protection from moisture, light, and repeated freeze–thaw cycles.
- Spill response and waste disposal: Manage spills, unused material, and consumables using procedures consistent with institutional chemical and biological waste policies.
- Documentation and traceability: Maintain accurate batch records, Certificates of Analysis (COAs), and internal logs to support reproducibility, traceability, and audit readiness.
Bluum Peptides supplies IGF-1 LR3 as a high-purity, lyophilized research compound and does not make therapeutic or medical claims. This compound is provided strictly for laboratory research use only and is not approved for clinical, diagnostic, or human application.
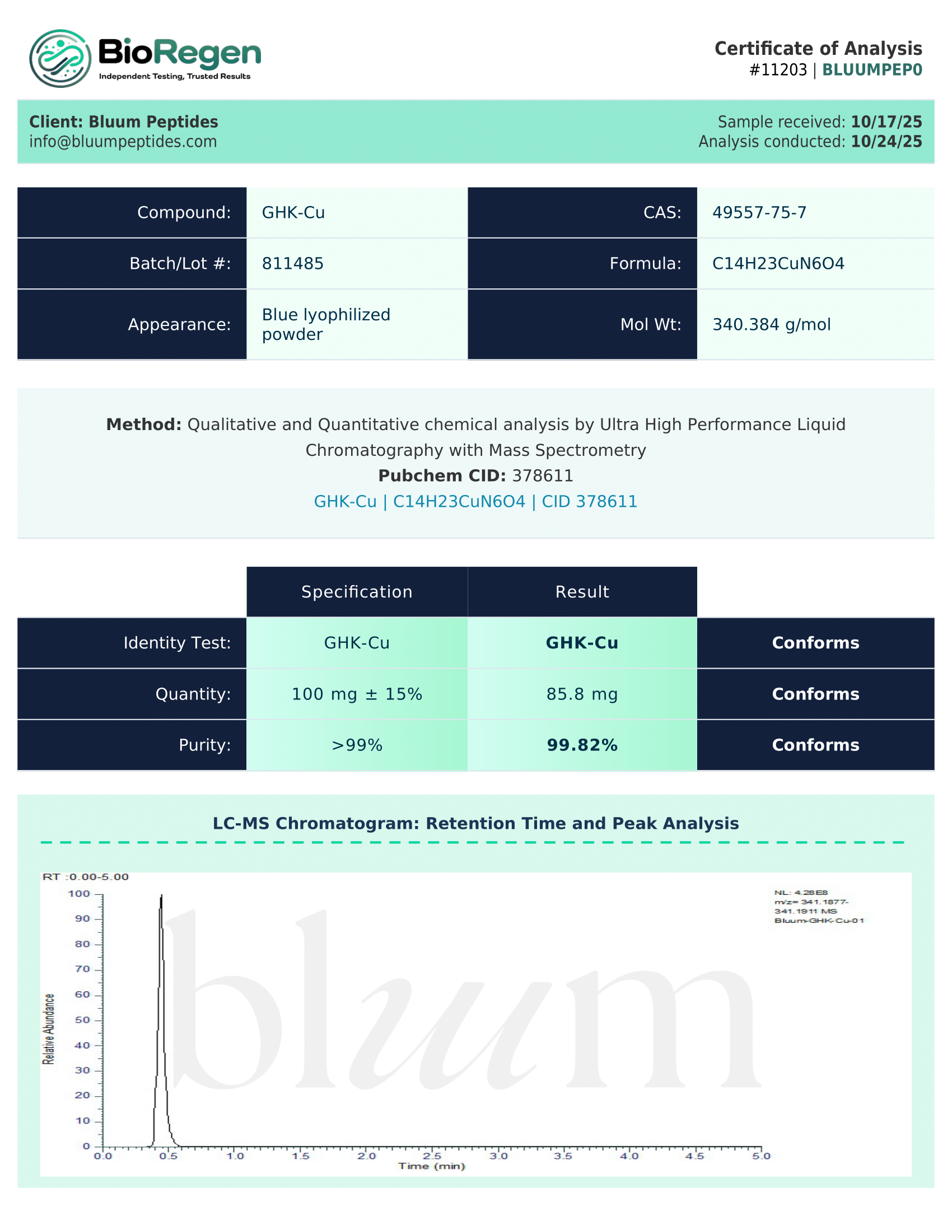
Certificate of Analysis (COA) & Quality Assurance
Each batch of IGF-1 LR3 supplied by Bluum Peptides is accompanied by a third-party–verified Certificate of Analysis (COA). These documents are provided to support research reproducibility, experimental transparency, and data integrity for researchers and institutions working with this modified IGF-1 analog.
For IGF-1 LR3, COAs typically include compound-relevant analytical data such as:
- Identity verification: Confirmation of the IGF-1 LR3 molecular structure using techniques such as mass spectrometry (MS) and/or NMR, appropriate for distinguishing modified IGF-1 analogs from native sequences.
- Purity analysis: Quantitative purity assessment, commonly via HPLC or comparable chromatographic methods, to support consistency in receptor signaling and growth-factor pathway studies.
- Peptide-specific characteristics: Reported molecular weight and sequence confirmation aligned with the extended and substituted structure of IGF-1 LR3.
- Physicochemical information: Available data related to solubility, concentration, or stability under defined laboratory testing conditions.
- Lot-specific documentation: Unique lot number, testing date, and analytical methodology summaries to support traceability and reproducibility.
Bluum Peptides works with independent, qualified analytical laboratories to perform third-party testing of IGF-1 LR3. This external verification process helps ensure objective assessment and consistent quality standards across production batches.
Certificates of Analysis are available in PDF format for review or upon request prior to purchase. Researchers are encouraged to retain COAs and associated batch records for internal audits, reproducibility documentation, and independent verification in accordance with institutional research protocols.
Scientific References
1. Assefa B, Mahmoud AM, Pfeiffer AFH, Birkenfeld AL, Spranger J, Arafat AM. Insulin-Like Growth Factor (IGF) Binding Protein-2, Independently of IGF-1, Induces GLUT-4 Translocation and Glucose Uptake in 3T3-L1 Adipocytes. Oxid Med Cell Longev. 2017;2017:3035184.
https://pmc.ncbi.nlm.nih.gov/articles/PMC5750484/
2. Alicia White, Jane Stremming, Stephanie R. Wesolowski, Saif I. Al-Juboori, Evgenia Dobrinskikh, Sean W. Limesand, Laura D. Brown, and Paul J. Rozance
American Journal of Physiology-Endocrinology and Metabolism 2025 328:1, E116-E125
https://journals.physiology.org/doi/full/10.1152/ajpendo.00259.2024
3. Baxter RC. Signaling Pathways of the Insulin-like Growth Factor Binding Proteins. Endocr Rev. 2023 Sep 15;44(5):753-778.
https://pmc.ncbi.nlm.nih.gov/articles/PMC10502586/
4. Khan MZ, Zugaza JL, Torres Aleman I. The signaling landscape of insulin-like growth factor 1. J Biol Chem. 2025 Jan;301(1):108047.
https://pmc.ncbi.nlm.nih.gov/articles/PMC11748690/
5. Biadgo B, Tamir W, Ambachew S. Insulin-like Growth Factor and its Therapeutic Potential for Diabetes Complications - Mechanisms and Metabolic Links: A Review. Rev Diabet Stud. 2020;16(1):24-34.
https://pmc.ncbi.nlm.nih.gov/articles/PMC9380093/
6. Wang, M., Zhang, J., Li, H., Li, Y., & Li, Z. (2025). Insulin‑like growth factor‑1 (IGF‑1) empowering tendon regenerative therapies. Frontiers in Bioengineering and Biotechnology, 13.
7. Song YH, Song JL, Delafontaine P, Godard MP. The therapeutic potential of IGF-I in skeletal muscle repair. Trends Endocrinol Metab. 2013 Jun;24(6):310-9.
https://pmc.ncbi.nlm.nih.gov/articles/PMC3732824/
8. Hosseinī Khoramī, S. A., & Movahhedī, A. (2015). PI3K/AKT pathway in modulating glucose homeostasis and its alteration in diabetes. Annals of Medical and Biomedical Sciences, 1(2), 46–55.
http://www.ambs‑journal.co.uk/articles/8%20AMBS46‑55‑20151.pdf