What is Retatrutide?
Retatrutide (also known as LY3437943) is a synthetic peptide designed to mimic three key metabolic receptors: the glucagon-like peptide 1 receptor (GLP-1R), the glucose-dependent insulinotropic polypeptide receptor (GIPR), and the glucagon receptor (GCGR). As such, retatrutide is a triple agonist.
By engaging all three receptors, Retatrutide is intended to help regulate key aspects of metabolism, including insulin release, glucose levels, and appetite. In doing so, reta can potentially improve glycemic control and support weight management as shown in early non-clinical and rodent trials.
Retatrutide is available as a high-purity, lyophilized research compound in 15 mg and 30 mg vials.
For research use only - not for human or veterinary consumption or medical applications.
Retatrutide Mechanism of Action (Based on Research)
Retatrutide is a triple-receptor agonist synthesized to activate the metabolic receptors GLP-1R, GIPR, and GCGR simultaneously. As these hormones naturally modulate appetite regulation, glucose handling, and energy expenditure, the synthetic form is thought to mimic these effects.
Retatrutide has a major advantage because it targets all three metabolic domains, which are usually targeted separately by single agonists such as Semaglutide or Liraglutide, or dual-agonist formulations like Tirzepatide.
Structural and Chemical Basis
In technical terms, Retatrutide is a formulation of 39 amino acids with a C20 fatty-diacid side chain that binds albumin, extending its half-life to roughly six days. This modification allows for once-weekly subcutaneous dosing and sustained receptor activation in research models.
GLP-1 Receptor (GLP-1R) Activation
From rodent research studies, activation of GLP-1R enhances glucose-dependent insulin secretion, helping maintain post-meal glucose control [1]. GLP-1R signaling also suppresses glucagon release, which limits hepatic glucose output.
These studies also suggest that GLP-1R slows gastric emptying, reducing the rate of nutrient absorption and prolonging satiety. Centrally, GLP-1R engagement influences hypothalamic pathways involved in appetite regulation, contributing to decreased caloric intake[2].
GIP Receptor (GIPR) Activation
Studies suggest that GIPR activation amplifies insulin secretion in response to nutrient intake, complementing the insulinotropic effect of GLP-1[3]. GIPR stimulation may improve insulin sensitivity, support more efficient nutrient partitioning, and modulate adipose-tissue metabolism.
When combined with GLP-1R activation, GIPR signaling appears to enhance the overall appetite-regulating and glucose-lowering effects observed with dual incretin therapy.
Glucagon Receptor (GCGR) Activation
Early rodent studies suggest that GCGR activation contributes a unique metabolic dimension by increasing energy expenditure and promoting fat oxidation. Through its hepatic actions, GCGR stimulation increases lipid mobilization, reduces fat storage, and modulates pathways involved in glucose and lipid metabolism[4].
Although glucagon signaling ordinarily raises blood glucose, this effect is counterbalanced in retatrutide by the robust insulinotropic actions of GLP-1R and GIPR, resulting in a metabolically favorable net profile.
Integrated Triple-Agonist Effect
By activating all three receptors together, retatrutide seemingly influences:
- Energy intake (reduced hunger, slower gastric emptying)
- Glucose handling (enhanced insulin secretion and sensitivity)
- Energy output (increased metabolic rate and fat oxidation)
This coordinated hormonal modulation may produce effects that exceed those of single- or dual-agonist peptides. Non-clinical studies have shown substantial weight reductions, improved glycemic control, reductions in liver fat, and broad improvements in cardiometabolic markers.
Researchers view retatrutide’s triple mechanism as a tool to explore metabolic and endocrine pathways not accessible through existing incretin-based therapies. Remember, BLUUM Peptides sells retatrutide for research use only and not for any clinical or therapeutic applications.
Research Applications (Observations from Studies)
Retatrutide is being actively examined in preclinical and clinical research settings for its effects on metabolism, body-weight regulation, glycemic control, and overall endocrine function. Findings described below reflect published research observations rather than established clinical outcomes.
Weight and Body Mass Regulation
Across phase 1 and phase 2 clinical studies, retatrutide has shown substantial effects on body weight, prompting interest in triple-agonist approaches to obesity research. In phase 2 trials, subjects receiving higher-dose regimens experienced average weight reductions approaching 24.2%[5].
These reductions exceeded the magnitude typically seen with single- or dual-agonist incretin therapies and occurred in a dose-dependent manner. Preclinical investigations similarly demonstrate reductions in adiposity, improved nutrient utilization, and favorable shifts in energy balance.
Metabolic and Glycemic Effects
Recent retatrutide trials have reported improvements in glycemic regulation, including reductions in fasting plasma glucose and HbA1c in individuals with impaired glucose metabolism[6].
Enhanced insulin sensitivity, improved postprandial glucose handling, and reductions in hepatic fat content have also been observed, including in studies examining metabolic-associated steatotic liver disease (MASLD).
Preclinical work supports these findings, showing enhanced insulin action, reduced hepatic lipid accumulation, and improved metabolic flexibility across tissues such as liver, muscle, and adipose tissue.
Cardiometabolic Markers
Early research has identified potential benefits across several cardiometabolic indicators, though these findings remain preliminary. Studies have documented favorable directional changes in blood lipids, blood pressure, and markers of hepatic steatosis, suggesting broader systemic effects beyond weight loss alone[6].
Investigators note that the combination of improved glycemic control, reduced adiposity, and increased energy expenditure may contribute to these trends, but long-term, large-scale trials are required to determine durability and clinical significance.
Please note that observed effects originate exclusively from controlled research environments - including animal models, early-phase clinical studies, and mechanistic trials.
Bluum Peptides does not claim therapeutic efficacy or health benefits.
Retatrutide vs Semaglutide vs Tirzepatide Comparison
|
Feature |
Semaglutide |
Tirzepatide |
Retatrutide |
|
Receptor Targets |
GLP-1 only |
GLP-1 + GIP |
GLP-1 + GIP + Glucagon |
|
Mechanism Complexity |
Single agonist |
Dual agonist |
Triple agonist |
|
Research Stage (for Retatrutide) / Approval Status |
Approved (Ozempic®, Wegovy®, and Rybelsus®) |
Approved (Mounjaro, Zepbound®) |
Research-use only |
|
Research Focus (for Retatrutide) |
— |
— |
Metabolic regulation, energy expenditure, glycemic & liver-fat studies |
|
Potential Research Advantages |
Well-characterized GLP-1 effects |
Adds GIP pathway modulation |
Adds glucagon pathway - broader metabolic modeling |
|
Disclaimer |
Prescription medication |
Prescription medication |
Research chemical - not for human use |
(Note: bluum does not sell Semaglutide or Tirzepatide - this table is provided for contextual comparison only.)
Laboratory Safety & Handling in Research Use
To support experimental integrity and repeatability, handle Retatrutide Peptide Blend using established laboratory best practices:
- Perform all handling using sterile technique and validated standard operating procedures (SOPs appropriate to the experimental model).
- Record lot numbers, storage conditions, preparation methods, and any reconstitution or dilution parameters in laboratory documentation.
- Retain certificates of analysis (COAs) and incoming quality control documentation alongside study records.
- Store, handle, and dispose of materials in accordance with institutional safety programs and the storage specifications provided.
- Maintaining thorough documentation and consistent handling protocols is essential for reproducibility across experiments and research sites.
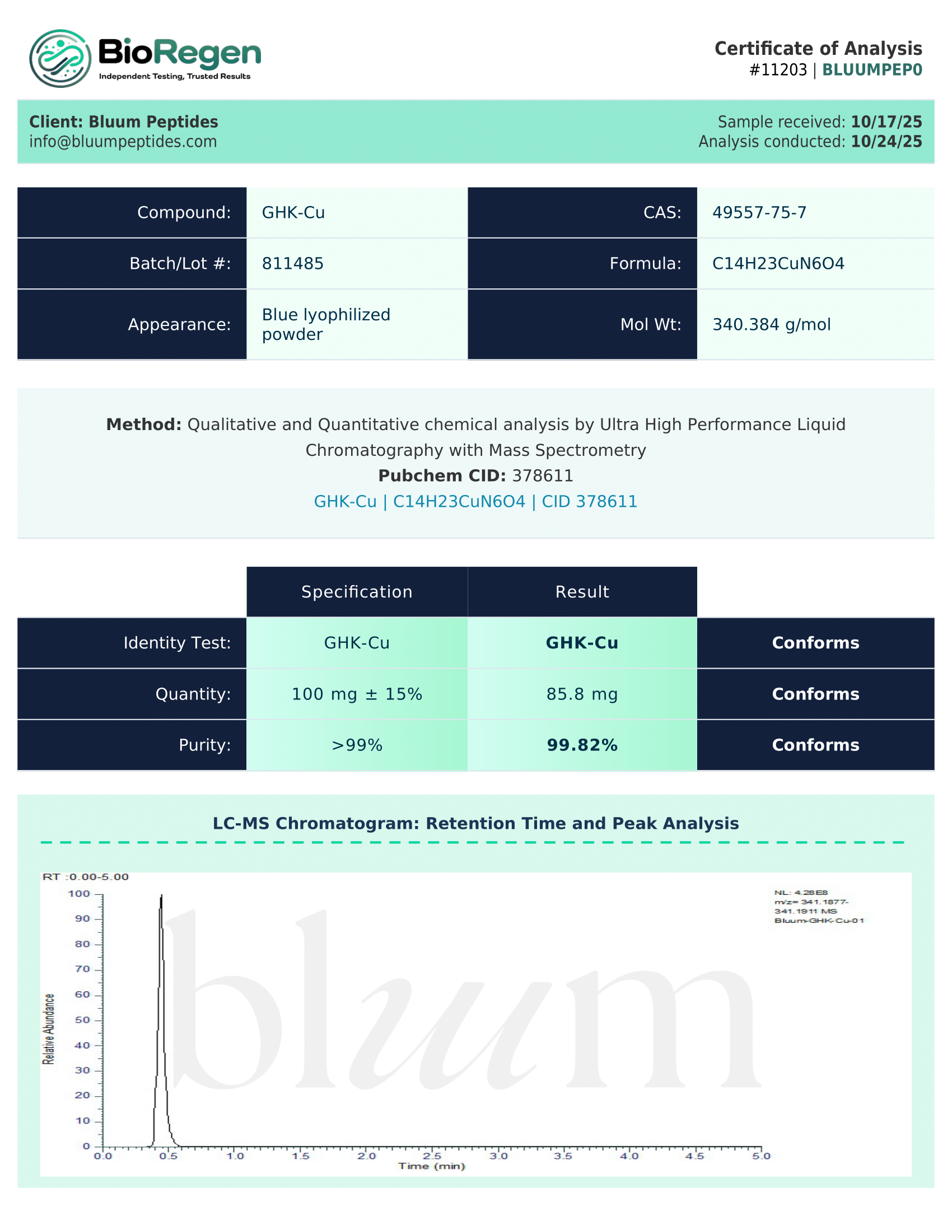
Certificate of Analysis (COA) & Quality Assurance
Each retatrutide batch is accompanied by a third-party–verified Certificate of Analysis (COA), ensuring transparent, research-grade quality control. The COA includes:
- Purity assessment (HPLC or reverse-phase chromatography)
- Mass spectrometry identity confirmation
- Solubility data and recommended buffers
- Lot number, testing date, and method documentation
Researchers may request sample COAs (PDF) ahead of purchase to confirm testing parameters, analytical methods, and batch consistency. You are encouraged to perform independent testing and verification.
Scientific References
- Zheng, Z., Zong, Y., Ma, Y. et al. Glucagon-like peptide-1 receptor: mechanisms and advances in therapy. Sig Transduct Target Ther 9, 234 (2024).
https://www.nature.com/articles/s41392-024-01931-z - Meloni AR, DeYoung MB, Lowe C, Parkes DG. GLP-1 receptor activated insulin secretion from pancreatic β-cells: mechanism and glucose dependence. Diabetes Obes Metab. 2013 Jan;15(1):15-27.
https://pmc.ncbi.nlm.nih.gov/articles/PMC3556522/ - James-Okoro, P.-P., Lewis, J. E., Gribble, F. M., & Reimann, F. (2025). The role of GIPR in food intake control. Frontiers in Endocrinology, 16.
https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2025.1532076/full - Villarroya F, Peyrou M, Giralt M. Adipose Tissue, at the Core of the Action of Incretin and Glucagon-Based Anti-Obesity Drugs. Curr Obes Rep. 2025 Sep 2;14(1):67.
https://pmc.ncbi.nlm.nih.gov/articles/PMC12405374/ - Goldney J, Hamza M, Surti F, Davies MJ, Papamargaritis D. Triple Agonism Based Therapies for Obesity. Curr Cardiovasc Risk Rep. 2025;19(1):18.
https://pmc.ncbi.nlm.nih.gov/articles/PMC12304053/ - Katsi V, Koutsopoulos G, Fragoulis C, Dimitriadis K, Tsioufis K. Retatrutide-A Game Changer in Obesity Pharmacotherapy. Biomolecules. 2025 May 30;15(6):796.
https://pmc.ncbi.nlm.nih.gov/articles/PMC12190491/ - Takrori E, Peshin S, Singal S. Gastrointestinal Adverse Effects of Anti-Obesity Medications in Non-Diabetic Adults: A Systematic Review. Medicina (Kaunas). 2025 Nov 5;61(11):1987.
https://pmc.ncbi.nlm.nih.gov/articles/PMC12654588/