What is GHK-Cu?
GHK-Cu (glycyl-L-histidyl-L-lysine copper complex) is a naturally occurring tripeptide–metal complex classified as a copper-binding signaling peptide. It was first identified in human plasma in the 1970s during studies examining age-related changes in peptide fractions, where it was observed to form a stable 1:1 coordination complex with copper ions.
Functionally, GHK-Cu is studied for its interactions with copper-dependent cellular signaling processes, with particular interest in transcriptional regulation, extracellular matrix dynamics, and cellular stress response pathways.
In the scientific literature, GHK-Cu is primarily investigated using in vitro systems and animal models to explore how copper-peptide complexes influence gene expression patterns and intracellular signaling behavior. These studies focus on mechanistic observations rather than outcomes, since controlled human clinical data remain limited. As a result, interpretation of findings should be restricted to a preclinical research framework.
GHK-Cu supplied by Bluum Peptides is provided as a high-purity, lyophilized research compound in 50mg and 100mg vial sizes, with quality verified by third-party labs to support research reliability. This compound is intended for research use only and is not for human or veterinary application.
GHK-Cu Mechanism of Action (Research Only)
GHK-Cu (glycyl-L-histidyl-L-lysine copper complex) is mainly studied as a pleiotropic signaling modulator. Its primary biological role in research models involves copper coordination and the downstream modulation of intracellular signaling, redox balance, and gene transcription[1]. Note that current mechanistic understanding is derived largely from in vitro experiments, cell culture systems, and animal studies, with no established clinical data supporting the same in humans.
Structural and Chemical Basis
GHK-Cu is a tripeptide composed of three amino acids, namely glycine, histidine, and lysine, forming a stable 1:1 coordination complex with a copper ion[1]. Chemically, it is classified as a peptide–metal complex rather than a modified or synthetic peptide analogue. The histidine residue provides strong metal-binding capacity, allowing copper to remain bioavailable while reducing free-ion reactivity in experimental systems.
This coordination structure is central to its research relevance. By acting as a copper carrier, GHK-Cu enables controlled interaction of copper with intracellular proteins, transcription factors, and redox-sensitive enzymes. In laboratory models, this stability supports reproducible observations related to cellular signaling and metal-dependent biochemical processes.
Modulation of Gene Expression and Transcriptional Activity
One of the most studied mechanisms of GHK-Cu in experimental settings involves its influence on gene expression. In cell-based models, exposure to the peptide–copper complex has been associated with broad shifts in transcriptional activity, particularly in genes linked to cellular structure, stress response, and protein turnover[2].
These effects are not attributed to a single receptor pathway but rather to indirect modulation of transcription factors and chromatin-associated processes.
Researchers interpret these findings as evidence of signaling modulation rather than direct activation. The observed gene expression patterns vary by cell type and experimental conditions, reinforcing that these mechanisms remain context-dependent and preclinical in nature.
Redox Signaling and Copper-Dependent Processes
GHK-Cu is also examined for its role in redox-sensitive signaling pathways due to copper’s involvement in oxidation–reduction reactions[3]. In vitro studies suggest the complex can influence cellular redox balance by regulating how copper participates in enzymatic systems, rather than allowing uncontrolled free-radical activity.
This controlled handling of copper is of interest in studies exploring oxidative stress, cellular resilience, and signaling adaptation. Importantly, these observations reflect biochemical behavior in isolated systems and animal models, not validated physiological outcomes.
Extracellular Matrix and Cellular Maintenance Pathways
Another research focus centers on how GHK-Cu interacts with pathways related to extracellular matrix organization and cellular maintenance. Experimental models have linked the complex to signaling cascades involved in protein synthesis, degradation, and structural organization within tissues[1]. These mechanisms are studied as part of broader investigations into how cells regulate their internal and external architecture.
Rather than producing defined effects, GHK-Cu is used as a tool to observe how copper-peptide signaling may influence coordinated cellular processes across multiple pathways.
Despite the links to regenerative research, GHK-Cu is not approved for any human or veterinary use. It is supplied by Bluum Peptides strictly for research use in copper-mediated signaling studies.
GHK-Cu Research Applications (Observations from Studies)
GHK-Cu is currently being studied for its role in copper-mediated cellular signaling. The observations below are drawn from in vitro experiments, animal models, and exploratory human studies, rather than established clinical outcomes. Therefore, these findings should be interpreted strictly within controlled experimental settings and are not intended to extend to human or veterinary use.
Gene Expression and Cellular Signaling Modulation
A primary research application of GHK-Cu involves its observed influence on gene expression patterns in cell-based and animal models[2]. Studies have reported broad transcriptional shifts affecting genes associated with cellular structure, stress response, and protein turnover.
Rather than acting through a single receptor, GHK-Cu appears to modulate signaling indirectly via copper-dependent transcription factors and regulatory enzymes.
Put more plainly, GHK-Cu allows researchers to model how copper handling influences gene regulation in living systems. This has made it a recurring point of interest in aging, tissue maintenance, and regeneration studies, where shifts in redox control and repair signaling are routinely observed. Results remain highly context-dependent, but the compound serves as a useful probe for studying regulatory stability at the cellular level.
Redox Balance and Oxidative Stress Pathways
GHK-Cu is also investigated in experimental models focused on redox signaling and oxidative stress. Copper plays a central role in oxidation–reduction reactions, and GHK-Cu is studied as a controlled copper carrier that may influence how cells manage reactive oxygen species[3].
Observations from in vitro systems suggest that GHK-Cu influences redox-sensitive signaling pathways rather than acting as a direct antioxidant. For researchers, this provides a way to examine how cells interpret and respond to oxidative challenges when copper is delivered in a peptide-bound form.
Such studies are commonly situated within aging, stress-response, and tissue maintenance models, where redox signaling plays a central regulatory role. Importantly, this work centers on pathway behavior and cellular adaptation, not on protective or restorative outcomes.
Extracellular Matrix and Tissue-Level Signaling Models
In tissue and organ-level research models, GHK-Cu has been examined for its association with extracellular matrix–related signaling pathways. Experimental observations include changes in markers linked to collagen organization, protease activity, and structural protein turnover[1].
These findings are frequently evaluated alongside models using unbound copper or non-chelated peptides to highlight how metal coordination alters downstream signaling behavior. In practical research terms, GHK-Cu functions as a probe for studying how cells integrate copper availability into pathways governing structural maintenance and remodeling—particularly in aging and tissue-turnover models.
The focus remains on understanding signaling coordination and regulatory logic, rather than generating predefined or outcome-driven tissue changes.
Comparative Research Context
Compared with single-target signaling molecules, GHK-Cu attracts research interest due to its multi-pathway, or pleiotropic, activity. Its effects are studied in contrast to free copper ions, which can be cytotoxic, and to peptides lacking metal-binding capacity. This comparative context helps researchers isolate how copper delivery method influences cellular responses.
Bluum Peptides implies no therapeutic or medical claims regarding GHK-Cu’s research applications. This compound is supplied strictly for research use only and is not intended for clinical, diagnostic, or human application.
GHK-Cu vs AHK-Cu vs KHK-Cu Comparison
|
Parameter |
GHK-Cu |
KHK-Cu |
|
|
Molecular / Structural Classification |
Tripeptide–metal coordination complex (glycine–histidine–lysine, copper-bound) |
Tripeptide–metal coordination complex (alanine–histidine–lysine, copper-bound) |
Tripeptide–metal coordination complex (lysine–histidine–lysine, copper-bound) |
|
Primary Biological Pathways Studied |
Copper-dependent signaling, broad transcriptional modulation, redox-responsive and extracellular matrix–associated pathways |
Copper-dependent signaling with emphasis on sequence-dependent gene expression and proliferative signaling |
Copper-dependent signaling examined primarily in redox-sensitive and transcriptional regulation models |
|
Mechanism Complexity |
Highly pleiotropic; influences multiple interconnected signaling and gene-regulatory pathways |
Moderately pleiotropic; overlaps with GHK-Cu but exhibits sequence-specific signaling bias |
More limited and model-dependent; used to probe how peptide sequence alters copper-mediated signaling |
|
Metal Handling |
Well-characterized copper chelation with controlled intracellular copper availability |
Similar copper chelation behavior with altered peptide–copper interaction dynamics |
Copper chelation with greater sensitivity to experimental conditions and peptide context |
|
Research Focus Areas |
Gene expression regulation, redox signaling, extracellular matrix remodeling, aging and tissue maintenance models |
Comparative studies evaluating how peptide sequence modifies copper-driven signaling outcomes |
Mechanistic comparison studies focused on sequence variation and copper coordination behavior |
|
Cellular / Systemic Scope |
Studied across intracellular signaling, tissue-level models, and age-associated research systems |
Primarily intracellular and tissue-model focused, often in side-by-side comparison with GHK-Cu |
Predominantly intracellular signaling and mechanistic copper-handling studies |
|
Comparative Research Value |
Serves as a reference compound for studying regulated copper signaling with reduced free-ion activity |
Useful for isolating sequence-driven differences in copper-mediated transcriptional responses |
Useful for probing how lysine-rich sequences influence copper coordination and signaling specificity |
|
Research Stage / Status |
Research compound. Not approved for human use. |
Research compound. Not approved for human use. |
Research compound. Not approved for human use. |
GHK-Cu Laboratory Safety & Handling (Research Use Only)
GHK-Cu is supplied strictly for laboratory research applications and should be handled only within controlled research environments by qualified personnel.
As a peptide–copper coordination complex intended for research use, GHK-Cu does not have a fully established toxicological or clinical safety profile. All safety considerations therefore apply exclusively to laboratory handling, material management, and risk mitigation practices, not to biological or clinical outcomes.
Handling considerations should reflect the compound’s chemical class and physical form (commonly supplied as a lyophilized peptide powder), as well as the specific preparation and experimental protocols employed. Reconstitution methods, concentration ranges, and downstream experimental use may influence appropriate handling controls, and institutional or protocol-specific guidance should always take precedence.
General laboratory safety and handling best practices include:
- Use sterile technique and adhere to institutional standard operating procedures (SOPs)
- Wear appropriate personal protective equipment (PPE), such as laboratory gloves, eye protection, and lab coats
- Use engineering controls (e.g., biosafety cabinets or fume hoods) to minimize aerosolization or accidental exposure during handling or reconstitution
- Store under defined environmental conditions appropriate to peptide stability, with protection from light, moisture, and temperature excursions
- Implement spill response and waste disposal procedures consistent with institutional, local, and regulatory requirements
- Maintain accurate documentation, including lot numbers, storage conditions, handling logs, Certificates of Analysis (COAs) and batch records
No definitive clinical safety data exist for GHK-Cu. Bluum Peptides supplies this compound exclusively for research use and does not provide medical, diagnostic, or therapeutic guidance.
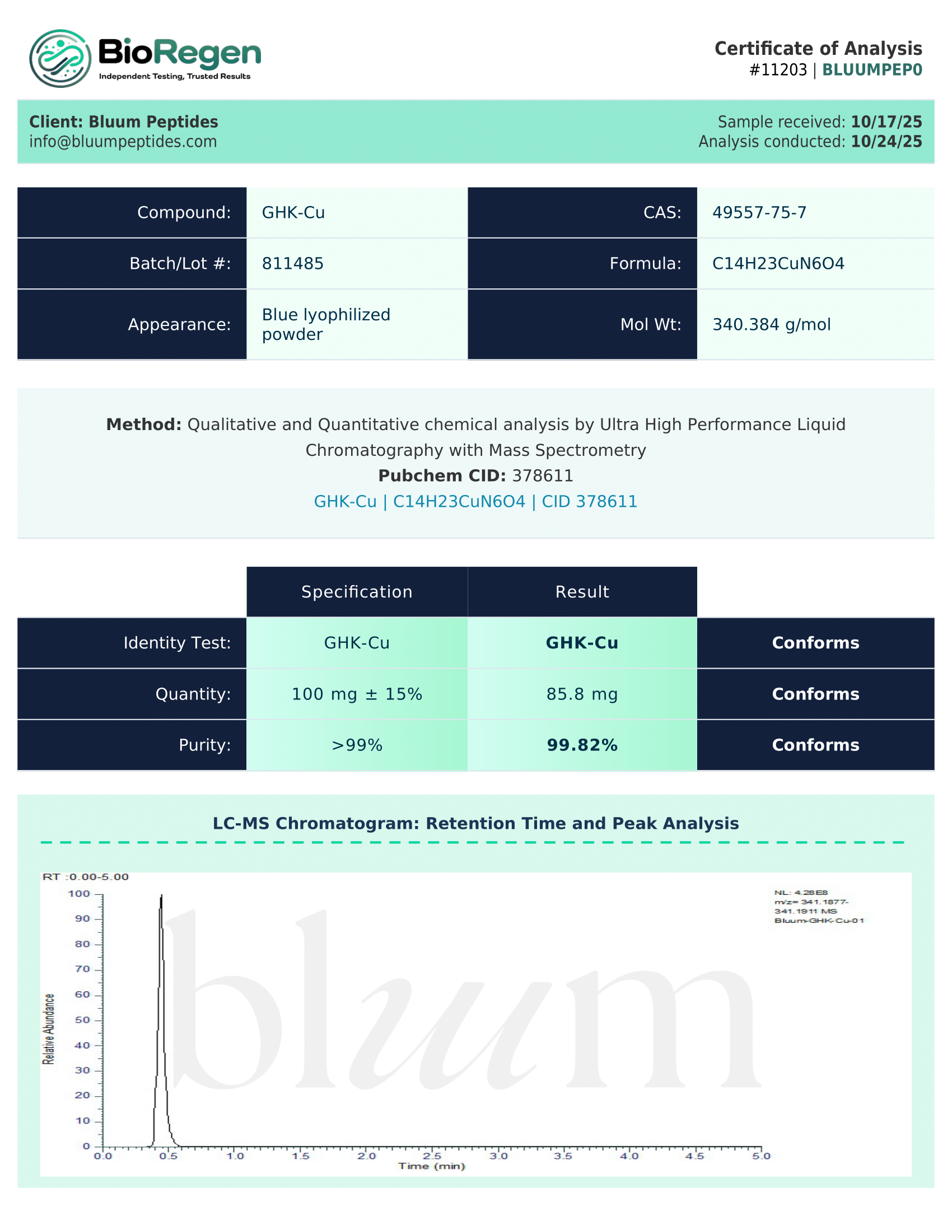
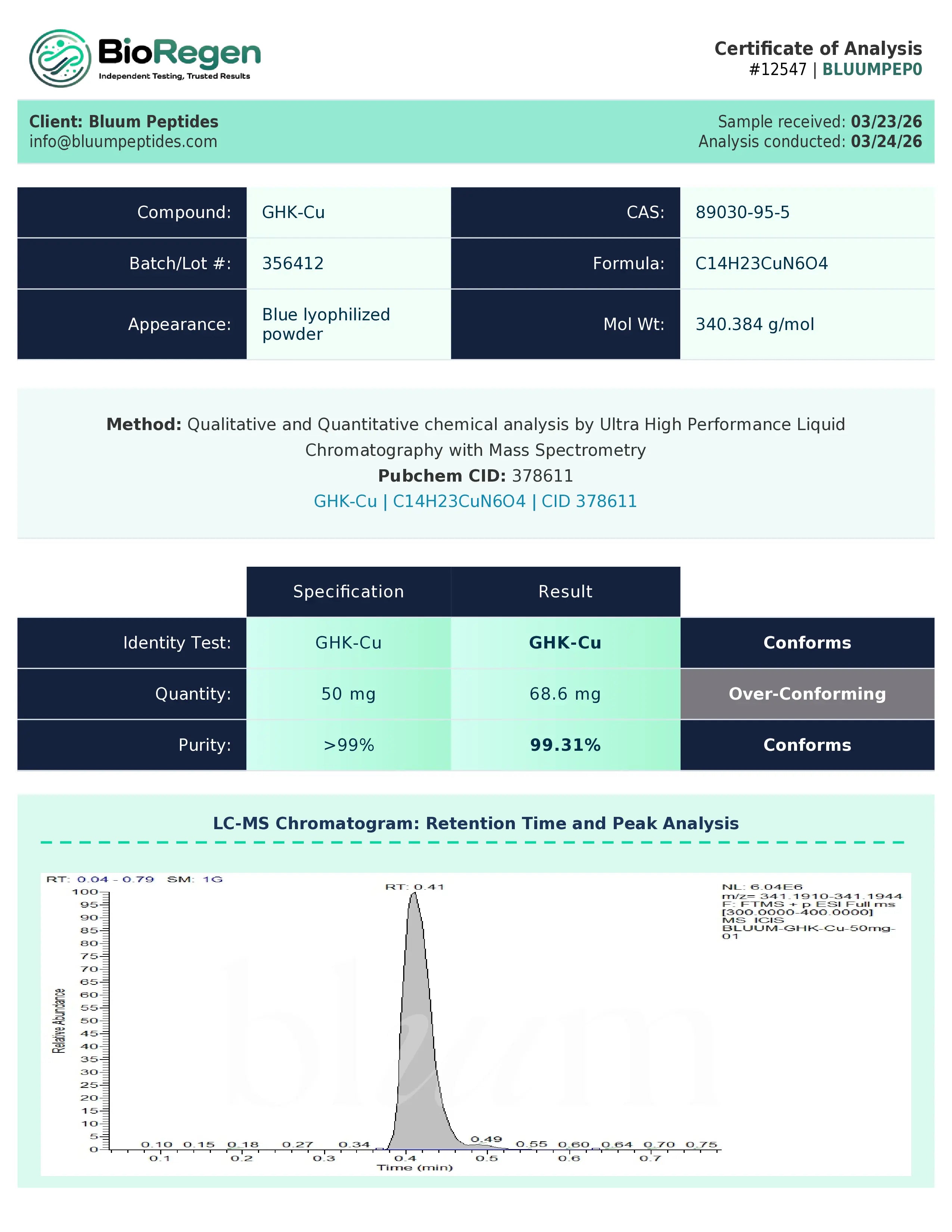
Certificate of Analysis (COA) & Quality Assurance
Each lot of GHK-Cu supplied by Bluum Peptides is accompanied by a third-party–verified Certificate of Analysis (COA) to support experimental reproducibility, traceability, and data integrity across research workflows.
COAs typically include analytical data appropriate to peptide–metal coordination complexes, such as:
- Identity verification using suitable analytical techniques (e.g., mass spectrometry, NMR, or equivalent methods)
- Purity or composition assessment using chromatography, HPLC, or assay-based analyses
- Relevant physicochemical information, which may include concentration, solubility characteristics, or stability-related data where applicable
- Lot number, testing date, and documentation of analytical methods used
Bluum Peptides works with independent analytical laboratories to provide objective verification and maintain consistent quality standards across product batches.
Certificates of Analysis are available for review or request in PDF format prior to purchase. Researchers are encouraged to retain COA documentation for audits, reproducibility assessments, or independent verification in accordance with institutional and regulatory requirements.
Scientific References
1. Pickart L, Vasquez-Soltero JM, Margolina A. GHK Peptide as a Natural Modulator of Multiple Cellular Pathways in Skin Regeneration. Biomed Res Int. 2015;2015:648108.
https://pmc.ncbi.nlm.nih.gov/articles/PMC4508379/
2. Pickart L, Margolina A. Regenerative and Protective Actions of the GHK-Cu Peptide in the Light of the New Gene Data. Int J Mol Sci. 2018 Jul 7;19(7):1987.
https://pmc.ncbi.nlm.nih.gov/articles/PMC6073405/
3. Pickart L, Vasquez-Soltero JM, Margolina A. The human tripeptide GHK-Cu in prevention of oxidative stress and degenerative conditions of aging: implications for cognitive health. Oxid Med Cell Longev. 2012;2012:324832.