What is Tesamorelin?
Tesamorelin is a synthetic peptide analog of growth hormone–releasing hormone (GHRH), structurally based on the endogenous GHRH (1–44) sequence and modified to improve stability for experimental use. It is classified as a single-pathway GHRH analog and is designed to interact selectively with the GHRH receptor (GHRHR), making it a useful tool for studying receptor-mediated signaling related to growth hormone regulation.
In the scientific literature, tesamorelin is primarily investigated in metabolic, endocrine, and cellular signaling research to better understand GHRH-driven pathways and downstream biochemical responses. Most findings to date originate from controlled laboratory settings, including in vitro systems, animal models, and limited exploratory human research, and should be interpreted within a preclinical framework.
Unlike multi-receptor peptides, tesamorelin’s receptor specificity allows researchers to examine targeted signaling mechanisms with reduced pathway overlap.
Bluum Peptides supplies tesamorelin as a high-purity, lyophilized research compound in 2mg, 5mg, and 10mg vials, with verified quality to support reproducible laboratory studies. Tesamorelin is currently available for research use only; not for human or veterinary application.
Tesamorelin Mechanism of Action (Research Only)
Tesamorelin is a synthetic peptide analogue designed to replicate the biological activity of endogenous growth hormone–releasing hormone (GHRH)[1]. In experimental systems, it functions primarily through receptor-mediated signaling, acting as a selective agonist of the GHRH receptor to modulate downstream endocrine and metabolic pathways.
Current understanding of tesamorelin’s mechanism of action is derived largely from in vitro experiments, animal models, and non-clinical research, rather than from comprehensive clinical mechanistic mapping.
Tesamorelin: Structural and Chemical Basis
Tesamorelin is a 44–amino acid synthetic peptide structurally based on human GHRH, with targeted amino acid substitutions that enhance resistance to enzymatic degradation[2]. These modifications increase its stability in experimental models, allowing for prolonged receptor engagement compared to native GHRH.
Chemically, tesamorelin is classified as a peptide hormone analogue. Its extended functional half-life supports sustained activation of downstream signaling cascades, making it a useful tool for studying growth hormone–related regulatory systems and time-dependent signaling dynamics.
GHRH Receptor Activation and Pituitary Signaling
In research models, tesamorelin binds to the GHRH receptor expressed on somatotroph cells, primarily within pituitary tissue cultures and animal systems. This receptor engagement activates G protein–coupled signaling pathways, most notably those involving cyclic AMP (cAMP) and protein kinase A (PKA)[3].
These signaling events stimulate transcriptional and secretory processes linked to growth hormone release. While this interaction is well-characterized at the cellular signaling level, observations are contextual to controlled experimental environments and do not imply predictable outcomes outside laboratory models.
Downstream Growth Hormone and IGF-1 Axis Modulation
Following receptor activation, tesamorelin indirectly influences the growth hormone–insulin-like growth factor 1 (IGF-1) axis[4]. Elevated growth hormone signaling observed in animal models leads to secondary modulation of IGF-1 synthesis in peripheral tissues.
This axis is often studied because it integrates signals related to growth, substrate utilization, and cellular turnover. From a mechanistic perspective, tesamorelin serves as a probe for examining how upstream hypothalamic signals propagate through endocrine feedback loops that coordinate energy availability and tissue-level signaling.
Metabolic and Cellular Signaling Interactions
Experimental data suggests that tesamorelin-associated signaling may influence pathways involved in lipid metabolism, glucose handling, and cellular energy allocation, primarily as downstream consequences of growth hormone activity[5]. In cellular and animal models, these pathways intersect with signaling networks that regulate nutrient partitioning and stress adaptation.
However, rather than acting directly on metabolic enzymes, tesamorelin’s role appears to be regulatory, shaping broader signaling environments that affect how cells respond to energetic demands and environmental stressors in research settings.
Research Context and Use Limitation
Tesamorelin is strictly for laboratory research purposes and is valued as a tool for investigating GHRH receptor biology, endocrine signaling, and growth hormone–related regulatory mechanisms. It is not approved for clinical, therapeutic, diagnostic, or human use. All described mechanisms reflect observations from non-clinical research models rather than established medical applications.
Tesamorelin Research Applications (Observations from Studies)
Tesamorelin has been examined in preclinical, translational, and early-phase human research as a selective growth hormone–releasing hormone (GHRH) analog. The observations summarized below reflect findings from controlled experimental settings, including mechanistic studies, animal models, and phase 1–2 human trials.
These observations do not represent established clinical outcomes and should not be interpreted as applicable to human or veterinary use outside of research contexts.
Growth Hormone Axis and Endocrine Signaling
Research involving tesamorelin frequently focuses on its interaction with the GHRH receptor and subsequent modulation of growth hormone–related signaling cascades. Experimental studies have observed stimulated endogenous growth hormone release in controlled settings, allowing researchers to explore downstream endocrine responses and feedback mechanisms.
In simpler terms, tesamorelin is used as a research tool to study how precise activation of the GHRH receptor influences hormone signaling without engaging multiple receptor systems, which helps isolate pathway-specific effects compared to broader-acting peptides.
Body Composition and Lipid-Related Markers
In animal models and early-phase human research, tesamorelin has been studied for its association with shifts in body composition–related markers, including changes in fat distribution and lipid parameters. These observations are typically reported as directional changes rather than definitive outcomes and are explored to better understand how growth hormone signaling may influence lipid metabolism.
Compared with multi-pathway metabolic peptides, tesamorelin’s single-receptor mechanism makes it useful for examining growth hormone–linked effects without confounding activity at appetite or insulin-related receptors.
Metabolic and Glycemic Research Contexts
Tesamorelin has also been included in metabolic research investigating glucose handling and insulin-related signaling under controlled conditions. Studies have explored how GHRH-mediated growth hormone release may indirectly influence glycemic markers, energy utilization, and metabolic homeostasis.
In other words, researchers are interested in how growth hormone signaling intersects with broader metabolic processes, not as a treatment approach but as a way to better map interconnected endocrine pathways in experimental systems.
Systemic and Inflammatory Marker Exploration
Some research has examined tesamorelin in relation to systemic biomarkers, including inflammatory and cardiometabolic indicators, particularly in tightly controlled human studies.
These investigations aim to understand whether modulation of the growth hormone axis produces measurable downstream biochemical shifts. Such findings are considered exploratory and are primarily valuable for hypothesis generation rather than confirmation of clinical relevance.
Tesamorelin vs Sermorelin vs CJC-1295
|
Feature |
Tesamorelin |
||
|
Compound class |
Synthetic GHRH analog |
Synthetic GHRH fragment (1–29) |
Synthetic GHRH analog (no albumin-binding complex) |
|
Primary receptor target |
GHRH receptor (GHRHR) |
GHRH receptor (GHRHR) |
GHRH receptor (GHRHR) |
|
Mechanism complexity |
Single-pathway, receptor-specific |
Single-pathway, receptor-specific |
Single-pathway with extended signaling duration |
|
Structural characteristics |
Full-length GHRH (1–44) with stabilizing modifications |
Truncated GHRH fragment |
GHRH analog without drug affinity complex (DAC) modification |
|
Signaling duration (research context) |
Moderate, controlled activation |
Shorter-acting signaling |
Short to moderate signaling duration |
|
Primary research focus areas |
Growth hormone axis, metabolic and endocrine signaling |
Baseline GHRH signaling, comparative GH studies |
Short-acting GHRH signaling, comparative GH pulsatility studies |
|
Research stage / regulatory status |
FDA-approved drug; also supplied as research compound |
FDA-approved drug; also studied as research standard |
Investigational / research-use compound (no FDA approval) |
|
Investigative value |
Allows targeted study of GHRH-mediated pathways with improved stability |
Useful as a reference GHRH fragment in mechanistic studies |
Useful for studying transient GHRH receptor activation and pulse-based signaling without extended exposure |
|
Intended use classification |
Prescription medication; research use only when supplied by Bluum Peptides |
Prescription medication; research standard in labs |
Research use only; not approved |
Tesamorelin Laboratory Safety & Handling in Research Use
Tesamorelin is a synthetic peptide supplied exclusively for controlled laboratory research and does not have a fully characterized toxicological or clinical safety profile. All safety considerations should therefore be evaluated within non-clinical research environments and interpreted in the context of experimental handling rather than human or veterinary exposure.
To support experimental integrity and reproducibility, tesamorelin should be handled using established laboratory best practices. This includes working under sterile conditions consistent with institutional standard operating procedures, documenting lot numbers, storage conditions, and preparation parameters, and retaining certificates of analysis alongside study records.
Maintaining consistent handling protocols across experiments is essential for traceability and data comparability.
Bluum Peptides makes no medical or therapeutic claims. Tesamorelin is supplied strictly for research use only and is not intended for clinical, diagnostic, human, or veterinary application.
- Sterile handling: Work under sterile conditions following institutional standard operating procedures (SOPs).
- Documentation: Record lot numbers, storage conditions, preparation methods, and retain certificates of analysis (COAs) for traceability.
- Personal protective equipment (PPE): Use lab coat, nitrile gloves, and eye protection; respiratory protection as required by facility guidelines.
- Engineering controls: Minimize aerosol or dust generation; perform weighing, transfer, or reconstitution in accordance with institutional safety protocols.
- Exposure prevention: Avoid inhalation, ingestion, or contact with skin and eyes; wash hands thoroughly after handling.
- Storage: Keep containers tightly sealed, protected from light and moisture; store at sub-zero temperatures and minimize repeated freeze–thaw cycles.
- Spill response and disposal: Follow institutional spill-response procedures and dispose of waste according to hazardous waste regulations.
- Reproducibility: Consistent handling protocols support experimental integrity and comparability across studies.
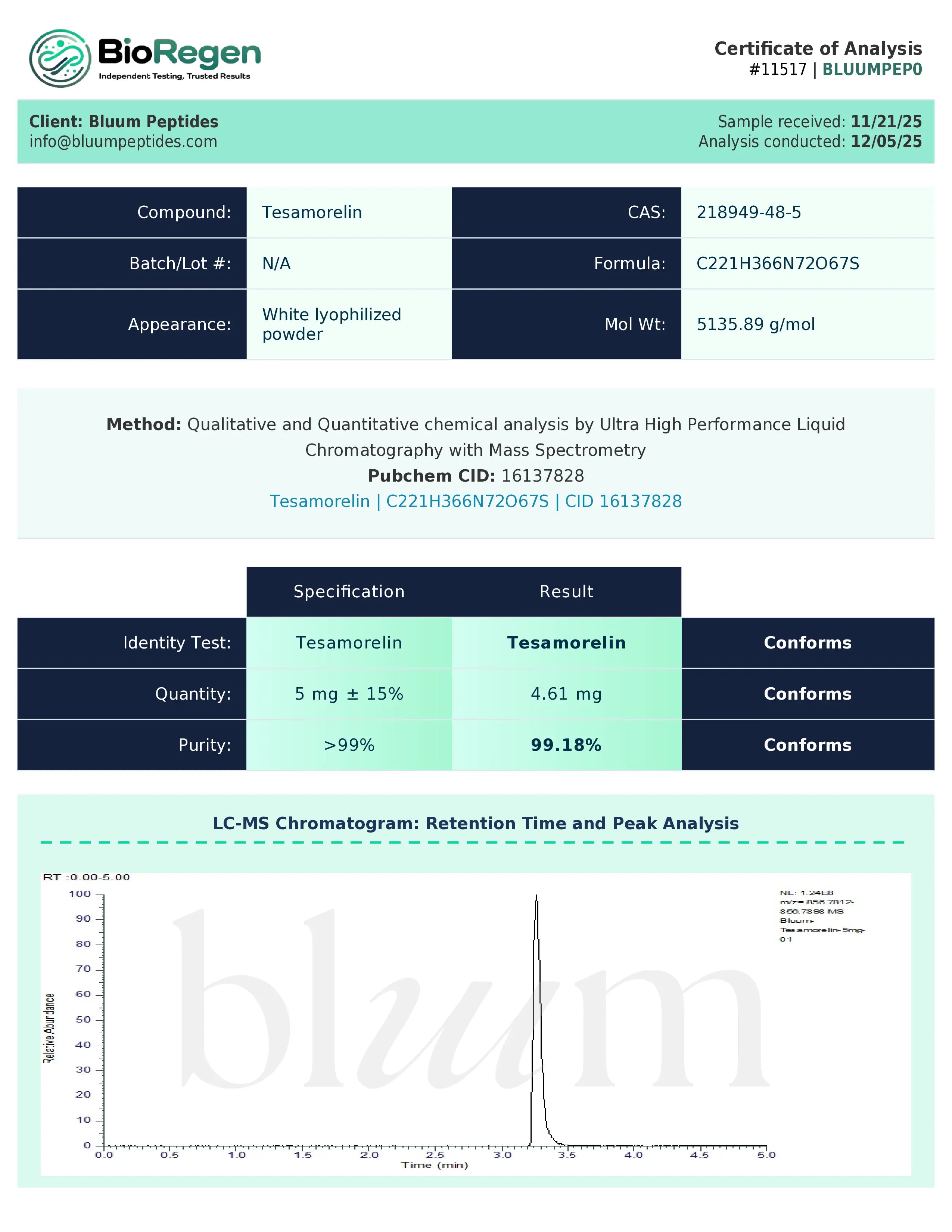
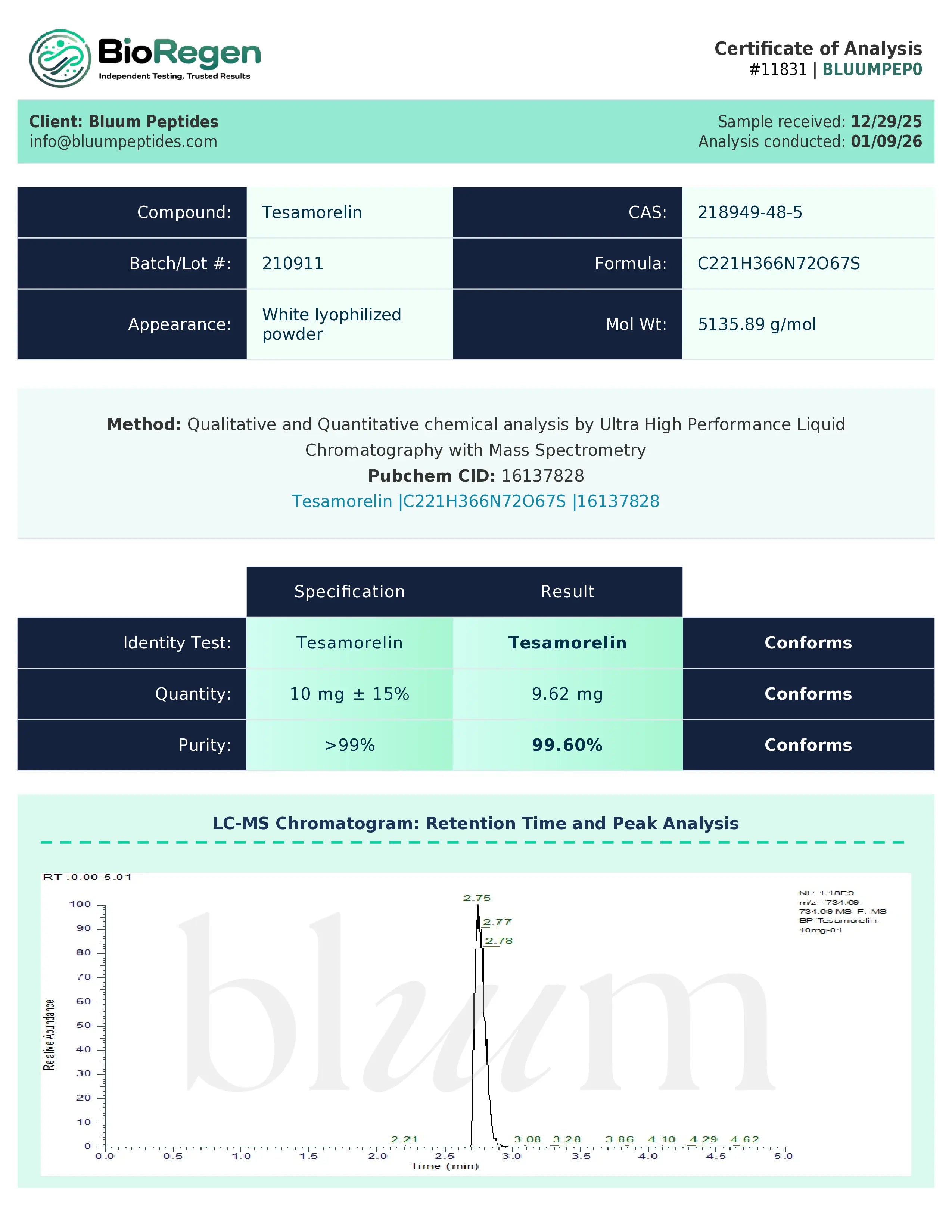
Certificate of Analysis (COA) & Quality Assurance
Bluum Peptides supplies each research-grade compound with a lot-specific, third-party–verified Certificate of Analysis (COA) to support experimental reproducibility and data integrity. The COA serves as a standardized quality document that allows researchers to independently verify analytical characteristics relevant to non-clinical study design and documentation.
COAs typically include:
- Identity verification using appropriate analytical techniques such as mass spectrometry, NMR, or equivalent methods suitable for the compound class
- Purity or composition as assessed through validated approaches including HPLC, chromatography, or assay-based analyses, depending on the material
- Where applicable, supporting physicochemical data such as concentration, solubility parameters, or stability observations in research conditions
- Associated lot number, testing date, and analytical methodologies used to ensure traceability
Quality assurance is supported through collaboration with independent analytical laboratories to provide objective verification and consistent testing standards. This third-party approach helps reduce bias and ensures that reported results reflect measured analytical outcomes rather than internal estimates.
Certificates of Analysis are available in PDF format for review or upon request prior to purchase. Researchers are encouraged to retain all COA documentation for internal records, audits, reproducibility assessments, or independent verification in accordance with institutional and laboratory protocols.
Scientific References
1. Adrian S, Scherzinger A, Sanyal A, Lake JE, Falutz J, Dubé MP, Stanley T, Grinspoon S, Mamputu JC, Marsolais C, Brown TT, Erlandson KM. The Growth Hormone Releasing Hormone Analogue, Tesamorelin, Decreases Muscle Fat and Increases Muscle Area in Adults with HIV. J Frailty Aging. 2019;8(3):154-159.
https://pmc.ncbi.nlm.nih.gov/articles/PMC6766405/
2. National Institute of Diabetes and Digestive and Kidney Diseases. (2012–). Tesamorelin. In LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. National Library of Medicine (US). Retrieved from https://www.ncbi.nlm.nih.gov/books/NBK548730/
3. Growth-Hormone-Releasing Hormone. (n.d.). In ScienceDirect Topics. Elsevier. Retrieved January 6, 2026, from https://www.sciencedirect.com/topics/neuroscience/growth-hormone-releasing-hormone
4. Yuen KCJ, Hjortebjerg R, Ganeshalingam AA, Clemmons DR, Frystyk J. Growth hormone/insulin-like growth factor I axis in health and disease states: an update on the role of intra-portal insulin. Front Endocrinol (Lausanne). 2024 Nov 21;15:1456195.
https://pmc.ncbi.nlm.nih.gov/articles/PMC11632222/
5. Clemmons DR, Miller S, Mamputu JC. Safety and metabolic effects of tesamorelin, a growth hormone-releasing factor analogue, in patients with type 2 diabetes: A randomized, placebo-controlled trial. PLoS One. 2017 Jun 15;12(6):e0179538.
https://pmc.ncbi.nlm.nih.gov/articles/PMC5472315/